- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Ornithology Journal
(Discontinued)
ISSN: 1874-4532 ― Volume 13, 2020
Painted Redstarts (Myioborus Pictus) Attack Larger Prey when Using Flush-Pursue Strategy
Piotr G. Jablonski1, 2, Sang-im Lee1, 3, *
Abstract
Introduction:
Prey escape reaction in insects is an antipredatory adaptation that is mediated by prey neural escape circuits with specific sensory properties.
Methods:
Certain insectivorous birds, flush-pursuers, exploit this visual sensitivity by employing conspicuous pivoting movements of spread tail and wings to flush the prey into the air where it is available for chase in aerial pursuits. Although it is known that this strategy increases the number of insects attacked, no information has been published on the size distribution of arthropods attacked using flush-pursue strategy vs. traditional gleaning and pecking off substrate strategy.
Results:
Based on one season of observational data of foraging redstarts (Myioborus pictus) we show that prey items that were flushed and chased were on average larger than prey pecked off of substrates.
Conclusion:
This may be one of the benefits from flush-pursue foraging – a strategy that is probably costly in terms of energy demands.
Article Information
Identifiers and Pagination:
Year: 2018Volume: 11
First Page: 34
Last Page: 38
Publisher Id: TOOENIJ-11-34
DOI: 10.2174/1874453201811010034
Article History:
Received Date: 10/11/2017Revision Received Date: 20/02/2018
Acceptance Date: 14/3/2018
Electronic publication date: 30/03/2018
Collection year: 2018
open-access license: This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International Public License (CC-BY 4.0), a copy of which is available at: (https://creativecommons.org/licenses/by/4.0/legalcode). This license permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
* Address correspondence to this author at the School of Undergraduate Studies, DGIST, Daegu 42988, South Korea, Tel: 82 53 785 6613; E-mail: sangim@dgist.ac.kr
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 10-11-2017 |
Original Manuscript | Painted Redstarts (Myioborus Pictus) Attack Larger Prey when Using Flush-Pursue Strategy | |
1. INTRODUCTION
Some species of insectivorous birds, the flush-pursuers (sensu) [1Remsen JV, Robinson SK. A classification scheme for foraging behavior of birds in terrestrial habitats. Stud Avian Biol 1990; 13: 144-60.], use conspicuous pivoting movements of spread tail and wings to exploit prey antipredatory escape reactions [2Edmunds M. Defence in Animals: A survey of anti-predator defences 1974., 3Evans DL, Schmidt JO. Insect defenses: Adaptive mechanisms and strategies of prey and predators 1990.]. These birds make a cryptic prey item conspicuous by flushing it into the air where it can be captured following an aerial pursuit, and by using this foraging method they increase the number of prey items delivered to their nestlings [4Barber MB, Barber DR, Jablonski PG. Painted redstart (Myioborus pictus). In: Poole A, Gill F, Eds. The Birds of North America, No 528 2000.-14Sherry TW, Homes RT. American Redstart (Setophaga ruticilla). In: Poole A, Gill F, Eds. Birds of North America, No 227, The Academy of Natural Sciences, Philadelphia, PA, and the American Ornithologists Union, Washington DC: 1997].
The painted redstart (Myioborus pictus) is one of such flush-pursuing species and it uses the conspicuous wing and tail displays during foraging [8Jablonski PG. A rare predator exploits prey escape behavior: The role of tail-fanning and plumage contrast in foraging of the painted redstart (Myioborus pictus). Behav Ecol 1999; 10: 7-14.
[http://dx.doi.org/10.1093/beheco/10.1.7] , 15Jabłonski PG. Sensory exploitation of prey: Manipulation of the initial direction of prey escapes by a conspicuous “rare enemy”. Proc Biol Sci 2001; 268(1471): 1017-22.
[http://dx.doi.org/10.1098/rspb.2001.1623] [PMID: 11375085] -17Jabłoński PG, Strausfeld NJ. Exploitation of an ancient escape circuit by an avian predator: Relationships between taxon-specific prey escape circuits and the sensitivity to visual cues from the predator. Brain Behav Evol 2001; 58(4): 218-40.
[http://dx.doi.org/10.1159/000057565] [PMID: 11964498] ]. When Myioborus redstarts use their displays, prey-pursuit flights are more frequent then they are during non-display-foraging [8Jablonski PG. A rare predator exploits prey escape behavior: The role of tail-fanning and plumage contrast in foraging of the painted redstart (Myioborus pictus). Behav Ecol 1999; 10: 7-14.
[http://dx.doi.org/10.1093/beheco/10.1.7] ] and they are able to feed their nestlings more frequently [8Jablonski PG. A rare predator exploits prey escape behavior: The role of tail-fanning and plumage contrast in foraging of the painted redstart (Myioborus pictus). Behav Ecol 1999; 10: 7-14.
[http://dx.doi.org/10.1093/beheco/10.1.7] , 10Mumme RL. Scare tactics in a neotropical warbler: white tail feathers enhance flush pursuit foraging performance in the slate-throated redstart (Myioborus miniatus). Auk 2002; 119: 1024-35., 18Jabłoński PG, Lasater K, Mumme RL, et al. Habitat-specific sensory-exploitative signals in birds: Propensity of dipteran prey to cause evolution of plumage variation in flush-pursuit insectivores. Evolution 2006; 60(12): 2633-42.
[http://dx.doi.org/10.1554/06-363.1] [PMID: 17263122] , 19Mumme RL, Galatowitsch ML, Jabłoński PG, Stawarczyk TM, Cygan JP. Evolutionary significance of geographic variation in a plumage-based foraging adaptation: An experimental test in the slate-throated redstart (Myioborus miniatus). Evolution 2006; 60(5): 1086-97.
[http://dx.doi.org/10.1111/j.0014-3820.2006.tb01185.x] [PMID: 16817547] ]. Although it is known that this strategy increases the number of insects attacked, no information has been published on the size distribution of arthropods attacked using flush-pursue strategy vs. traditional gleaning and pecking off substrate strategy. We ask here whether prey items that were flushed and chased differ in size from those that were pecked off of substrates. The answer will expand our knowledge about predator-prey interactions in the systems with flush-pursue predators.
2. METHODS
The research was conducted in the Cave Creek Canyon, Chiricahua Mountains, near the Southwestern Research Station, Portal, AZ (latitude: 31.866; longitude: 109.238; UTM zone 12). During the breeding season of 1997 (May - June) foraging redstarts were followed to describe food-size distributions for birds during aerial chasing or pecking off the prey. During field work concerning breeding phenology, singing and territorial behavior we opportunistically collected foraging observations of birds across the whole study area. Although many birds were not individually banded, foraging observations were sparsely distributed in space and time and we assumed that they were independent from each other and probably from 24 different birds. The observations were conducted along the Cave Creek with its tributaries and along the East Turkey Creek, altogether comprising the total length of about 17 km. Redstart breeding territories and nests were located along these creeks (Fig. 1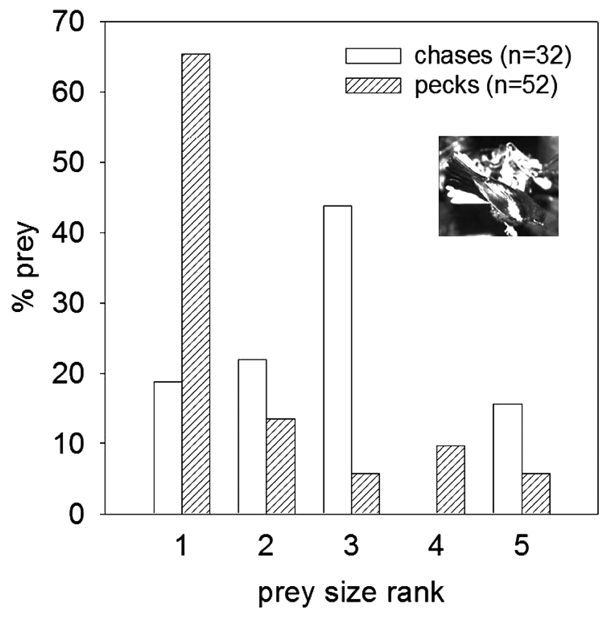 ) (nest locations from 1997). When a foraging bird was spotted the observer attempted to watch it closely through binoculars while audio-recording the observations for later translation. Only occasionally, it was possible to determine the size of the prey captured by a foraging bird. In total, we collected such information for 32 prey items (average 1.3 per bird) captured during aerial chases after flushed prey and for 52 prey items (average 2.2 per bird) captured by picking off the substrate (trunks, branches and leaves). Because the determination of size is not always possible the foraging sequences in which it was achieved several times for one bird were interspersed in time and across different trees. Therefore we regarded them as independent events. We coded prey size captured by birds in five size ranks: 1. prey smaller than 2mm; 2. prey larger than 2 mm and smaller than or equal to half of redstart bill length; 3. prey larger than half but smaller than or equal to one bill length; 4. prey larger than one bill length and smaller than or equal to 1.5 bill lengths; 5. prey larger than 1.5 bill lengths. Using a Mann-Whitney U test, we tested the null hypothesis that median prey size rank does not differ between the two hunting techniques: pecking off the substrate and chasing prey in air. Using G-test, we also compared distributions of the prey sizes between these two hunting techniques. We used sequential-Bonferroni correction of significance levels for these two tests (k=2; Rice, 1989). The taxonomic category of the prey was difficult to ascertain in most cases.
) (nest locations from 1997). When a foraging bird was spotted the observer attempted to watch it closely through binoculars while audio-recording the observations for later translation. Only occasionally, it was possible to determine the size of the prey captured by a foraging bird. In total, we collected such information for 32 prey items (average 1.3 per bird) captured during aerial chases after flushed prey and for 52 prey items (average 2.2 per bird) captured by picking off the substrate (trunks, branches and leaves). Because the determination of size is not always possible the foraging sequences in which it was achieved several times for one bird were interspersed in time and across different trees. Therefore we regarded them as independent events. We coded prey size captured by birds in five size ranks: 1. prey smaller than 2mm; 2. prey larger than 2 mm and smaller than or equal to half of redstart bill length; 3. prey larger than half but smaller than or equal to one bill length; 4. prey larger than one bill length and smaller than or equal to 1.5 bill lengths; 5. prey larger than 1.5 bill lengths. Using a Mann-Whitney U test, we tested the null hypothesis that median prey size rank does not differ between the two hunting techniques: pecking off the substrate and chasing prey in air. Using G-test, we also compared distributions of the prey sizes between these two hunting techniques. We used sequential-Bonferroni correction of significance levels for these two tests (k=2; Rice, 1989). The taxonomic category of the prey was difficult to ascertain in most cases.
3. RESULTS
The size rank of prey chased in air (mean, [median, (minimum-maximum)]; 2.6 [3Evans DL, Schmidt JO. Insect defenses: Adaptive mechanisms and strategies of prey and predators 1990.] (1-5), n=32) was larger (Mann Whitney test; Z1=2.68, P=0.008) than the size rank of prey pecked off the substrate (1.8) [1Remsen JV, Robinson SK. A classification scheme for foraging behavior of birds in terrestrial habitats. Stud Avian Biol 1990; 13: 144-60.] (1-5),(n=52), and the two prey size distributions differed between each other (Fig. 1 ); (G4 = 25.71), (P=0.00008). Most prey pecked off substrates consisted of small unrecognizable insects. Among 10 insects pecked that were recognizable there were 6 caterpillars, 3 spiders, and 1 cicada nymph. Among 8 insects chased that were recognizable there were 3 flies, 2 cicadas, 1 moth and 2 Hemipterans. Caterpillars were the largest prey items pecked off the substrate. The details of the diet of redstart’s nestlings during breeding season are going to be reported separately, but these taxonomic groups of prey are commonly brought to nestlings.
); (G4 = 25.71), (P=0.00008). Most prey pecked off substrates consisted of small unrecognizable insects. Among 10 insects pecked that were recognizable there were 6 caterpillars, 3 spiders, and 1 cicada nymph. Among 8 insects chased that were recognizable there were 3 flies, 2 cicadas, 1 moth and 2 Hemipterans. Caterpillars were the largest prey items pecked off the substrate. The details of the diet of redstart’s nestlings during breeding season are going to be reported separately, but these taxonomic groups of prey are commonly brought to nestlings.
4. DISCUSSION
In addition to previous analyses on foraging Painted redstarts and Slate-throated redstarts [8Jablonski PG. A rare predator exploits prey escape behavior: The role of tail-fanning and plumage contrast in foraging of the painted redstart (Myioborus pictus). Behav Ecol 1999; 10: 7-14.
[http://dx.doi.org/10.1093/beheco/10.1.7] , 10Mumme RL. Scare tactics in a neotropical warbler: white tail feathers enhance flush pursuit foraging performance in the slate-throated redstart (Myioborus miniatus). Auk 2002; 119: 1024-35., 19Mumme RL, Galatowitsch ML, Jabłoński PG, Stawarczyk TM, Cygan JP. Evolutionary significance of geographic variation in a plumage-based foraging adaptation: An experimental test in the slate-throated redstart (Myioborus miniatus). Evolution 2006; 60(5): 1086-97.
[http://dx.doi.org/10.1111/j.0014-3820.2006.tb01185.x] [PMID: 16817547] ], that indicated benefit from flush-pursuing owing to the larger number of attacks on prey, here we focused on the size of prey estimated in the field conditions. The recorded size distribution of prey suggested that Painted redstarts may additionally benefit because the flush-pursue strategy results in on average larger prey than the prey pecked off the substrates. Jablonski and Strausfeld [16Jablonski PG, Strausfeld NJ. Exploitation by a recent avian predator of an ancient arthropod escape circuit: Prey sensitivity and elements of the displays by predators. Brain Behav Evol 2000; 56: 94-106.
[http://dx.doi.org/10.1159/000006680] [PMID: 11111136] , 17Jabłoński PG, Strausfeld NJ. Exploitation of an ancient escape circuit by an avian predator: Relationships between taxon-specific prey escape circuits and the sensitivity to visual cues from the predator. Brain Behav Evol 2001; 58(4): 218-40.
[http://dx.doi.org/10.1159/000057565] [PMID: 11964498] ] and Jablonski et al. [18Jabłoński PG, Lasater K, Mumme RL, et al. Habitat-specific sensory-exploitative signals in birds: Propensity of dipteran prey to cause evolution of plumage variation in flush-pursuit insectivores. Evolution 2006; 60(12): 2633-42.
[http://dx.doi.org/10.1554/06-363.1] [PMID: 17263122] ] have shown that models of redstarts approaching a variety of potential prey at the present study site may evoke escape responses in a variety of medium- and large-sized prey such as flies, leafhoppers or moths. In another flush-pursuing species, the American Redstart (Setophaga ruticilla), Robinson & Holmes [11Robinson SK, Holmes RT. Foraging behavior of forest birds: The relationships among search tactics, diet, and habitat structure. Ecology 1982; 63: 1918-31.
[http://dx.doi.org/10.2307/1940130] ] and Keast et al. [9Keast A, Pearce L, Saunders S. How convergent is American Redstart (Setophaga ruticilla, Parulinae) with flycatchers (Tyrannidae) in morphology and feeding behavior? Auk 1995; 112: 310-25.
[http://dx.doi.org/10.2307/4088719] ] also suggested that the majority of prey pursued in flight was medium or large size, while the prey pecked off the substrate by warblers often contained small and energetically unrewarding insects (in addition to some large caterpillars during specific time of year). Similarly, flush-pursue strategy of tail-flicking in the Hooded Warbler (Setophaga citrina) results in different types of prey brought to the nestlings [20Mumme RL. White tail spots and tail flicking behavior enhance foraging performance in the Hooded Warbler. Auk 2014; 131: 141-9.
[http://dx.doi.org/10.1642/AUK-13-199.1] ]. Hence, it seems that regardless of whether flush-pursuers use tail-flicking or spread-tail pivoting the birds capture more and different, possibly more energy-rewarding, insects. It is possible that temperature may affect this difference in prey size between flush-pursuing and gleaning because it is possible that insects may flush more easily in higher temperatures. Future studies should address this issue.
CONCLUSION
In summary, this is the first report based on direct observations of foraging birds that documents how flush-pursue foraging method may not only increase the number of insects available for pursuit, but that those insects are of medium and large size, providing energetic benefits that may possibly help to offset the costs of aerial chases by flush-pursuers.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No human or animal were used in this study that on bases of this research.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
The field work was funded by the Maria Sklodowska-Curie Joint Fund II (PAN/EPA-96-241), the Kosciuszko Foundation, the American Museum of Natural History (Chapman Grants), the Association for the Study of Animal Behaviour, and the Committee for Scientific Research (KBN grant 6 PO4F 030 08). Analysis and manuscript preparation was supported by the KRF project nr. 0409-20080118, the NRF project 2016R1D1A1B03934340, the Brain Korea 21 Project, and the grant (3344-20080067) from the College of Natural Sciences, Seoul National University and by the DGIST Start-up Fund Program of the Ministry of Science, ICT and Future Planning (2018010100). The staff of the Southwestern Research Station - W.Sherbrooke, E. Sherbrooke, A. Oritz, L.Oritz, and S.Cox, as well as station volunteers are gratefully acknowledged.
REFERENCES
| [1] | Remsen JV, Robinson SK. A classification scheme for foraging behavior of birds in terrestrial habitats. Stud Avian Biol 1990; 13: 144-60. |
| [2] | Edmunds M. Defence in Animals: A survey of anti-predator defences 1974. |
| [3] | Evans DL, Schmidt JO. Insect defenses: Adaptive mechanisms and strategies of prey and predators 1990. |
| [4] | Barber MB, Barber DR, Jablonski PG. Painted redstart (Myioborus pictus). In: Poole A, Gill F, Eds. The Birds of North America, No 528 2000. |
| [5] | Fitzpatrick JW. Foraging behavior of neotropical tyrant flycatchers. Condor 1980; 82: 43-57. [http://dx.doi.org/10.2307/1366784] |
| [6] | Holmes RT, Recher HF. Search tactics of insectivorous birds foraging in an Australian eucalypt forest. Auk 1986; 103: 515-30. |
| [7] | Jablonski P. Adaptive significance of colour patterns in Painted Redstart. J Ornithol 1994; 135(Suppl.): 147. |
| [8] | Jablonski PG. A rare predator exploits prey escape behavior: The role of tail-fanning and plumage contrast in foraging of the painted redstart (Myioborus pictus). Behav Ecol 1999; 10: 7-14. [http://dx.doi.org/10.1093/beheco/10.1.7] |
| [9] | Keast A, Pearce L, Saunders S. How convergent is American Redstart (Setophaga ruticilla, Parulinae) with flycatchers (Tyrannidae) in morphology and feeding behavior? Auk 1995; 112: 310-25. [http://dx.doi.org/10.2307/4088719] |
| [10] | Mumme RL. Scare tactics in a neotropical warbler: white tail feathers enhance flush pursuit foraging performance in the slate-throated redstart (Myioborus miniatus). Auk 2002; 119: 1024-35. |
| [11] | Robinson SK, Holmes RT. Foraging behavior of forest birds: The relationships among search tactics, diet, and habitat structure. Ecology 1982; 63: 1918-31. [http://dx.doi.org/10.2307/1940130] |
| [12] | Sherry TW. Ecological and evolutionary inferences from morphology, foraging behavior, and diet of sympatric insectivorous neotropical flycatchers (Tyrannidae). PhD Thesis 1982. |
| [13] | Sherry TW. Comparative and dietary ecology of sympatric insectivorous neotropical flycatchers (Tyrannidae). Ecol Monogr 1984; 54: 313-38. [http://dx.doi.org/10.2307/1942500] |
| [14] | Sherry TW, Homes RT. American Redstart (Setophaga ruticilla). In: Poole A, Gill F, Eds. Birds of North America, No 227, The Academy of Natural Sciences, Philadelphia, PA, and the American Ornithologists Union, Washington DC: 1997 |
| [15] | Jabłonski PG. Sensory exploitation of prey: Manipulation of the initial direction of prey escapes by a conspicuous “rare enemy”. Proc Biol Sci 2001; 268(1471): 1017-22. [http://dx.doi.org/10.1098/rspb.2001.1623] [PMID: 11375085] |
| [16] | Jablonski PG, Strausfeld NJ. Exploitation by a recent avian predator of an ancient arthropod escape circuit: Prey sensitivity and elements of the displays by predators. Brain Behav Evol 2000; 56: 94-106. [http://dx.doi.org/10.1159/000006680] [PMID: 11111136] |
| [17] | Jabłoński PG, Strausfeld NJ. Exploitation of an ancient escape circuit by an avian predator: Relationships between taxon-specific prey escape circuits and the sensitivity to visual cues from the predator. Brain Behav Evol 2001; 58(4): 218-40. [http://dx.doi.org/10.1159/000057565] [PMID: 11964498] |
| [18] | Jabłoński PG, Lasater K, Mumme RL, et al. Habitat-specific sensory-exploitative signals in birds: Propensity of dipteran prey to cause evolution of plumage variation in flush-pursuit insectivores. Evolution 2006; 60(12): 2633-42. [http://dx.doi.org/10.1554/06-363.1] [PMID: 17263122] |
| [19] | Mumme RL, Galatowitsch ML, Jabłoński PG, Stawarczyk TM, Cygan JP. Evolutionary significance of geographic variation in a plumage-based foraging adaptation: An experimental test in the slate-throated redstart (Myioborus miniatus). Evolution 2006; 60(5): 1086-97. [http://dx.doi.org/10.1111/j.0014-3820.2006.tb01185.x] [PMID: 16817547] |
| [20] | Mumme RL. White tail spots and tail flicking behavior enhance foraging performance in the Hooded Warbler. Auk 2014; 131: 141-9. [http://dx.doi.org/10.1642/AUK-13-199.1] |




