- Home
- About Journals
-
Information for Authors/ReviewersEditorial Policies
Publication Fee
Publication Cycle - Process Flowchart
Online Manuscript Submission and Tracking System
Publishing Ethics and Rectitude
Authorship
Author Benefits
Reviewer Guidelines
Guest Editor Guidelines
Peer Review Workflow
Quick Track Option
Copyediting Services
Bentham Open Membership
Bentham Open Advisory Board
Archiving Policies
Fabricating and Stating False Information
Post Publication Discussions and Corrections
Editorial Management
Advertise With Us
Funding Agencies
Rate List
Kudos
General FAQs
Special Fee Waivers and Discounts
- Contact
- Help
- About Us
- Search

The Open Systems Biology Journal
(Discontinued)
ISSN: 1876-3928 ― Volume 5, 2014
Quartz Crystal Microbalance Immunoassay of Hepatitis B Surface Antigen Using Magnetic CoFe2O4/Cu2O Composite Nanoparticles as Immunosensing Probe
Feng Tian1, Dianping Tang*, 2
Abstract
This study describes a new strategy toward the development of advanced immunosensors based on chemically functionalized magnetic-core/porous-shell CoFe2O4/Cu2O composite nanoparticles, and the preparation, characterization, and measurement of relevant properties of the immunosensor useful for the detection of hepatitis B surface antigen (HBsAg) in clinical immunoassay. The immunosensor based on the combination of a magnetic nanocore and a Cu2O shell shows good adsorption properties for the attachment of hepatitis B surface antibody (HBsAb) selective to HBsAg. The core-shell nanostructure presents good magnetic properties to facilitate and modulate the way while it was integrated into a magnetic interface. Under optimal conditions, the resulting composite presents good frequency response for the detection of HBsAg, and allows detection of HBsAg at concentration as low as 0.5 ng/mL. Importantly, the proposed methodology could be extended to the detection of other antigens or biocompounds.
Article Information
Identifiers and Pagination:
Year: 2010Volume: 3
First Page: 29
Last Page: 31
Publisher Id: TOSYSBJ-3-29
DOI: 10.2174/1876392801003010029
Article History:
Received Date: 2/3/2010Revision Received Date: 22/4/2010
Acceptance Date: 22/4/2010
Electronic publication date: 6/5/2010
Collection year: 2010
open-access license: This is an open access article licensed under the terms of the Creative Commons Attribution Non-Commercial License (http: //creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted, non-commercial use, distribution and reproduction in any medium, provided the work is properly cited.
* Address correspondence to this author at the College of Chemistry and Chemical Engineering, Fuzhou University, Fuzhou, 350108, P.R. China; Tel: +86 591 2286 6125; Fax: +86 591 2286 6135; E-mail: dianping.tang@hotmail.com
| Open Peer Review Details | |||
|---|---|---|---|
| Manuscript submitted on 2-3-2010 |
Original Manuscript | Quartz Crystal Microbalance Immunoassay of Hepatitis B Surface Antigen Using Magnetic CoFe2O4/Cu2O Composite Nanoparticles as Immunosensing Probe | |
1. INTRODUCTION
The properties of two-dimensional assemblies of metal nanoparticles are controlled by the composition, geometry and spatial arrangement of the nanoparticle building blocks [1]. Protein-mediated assembly of nanoparticles is a potent tool for fabrication of new materials, which combine tunable nanoparticle features (size, surface functionality, and core properties) with the unique physical and chemical properties of protein and peptides [2]. Although great efforts have been focused on the assemblies of spherical or pseudospherical nanostructures, anisotropic metal nanomaterials are an intrinsically attractive class of building blocks for such assemblies due to their size- and shape-dependent properties [3]. A promising protocol would be to target the surface of the proteins through complementary interactions, using the shape and physical characteristics of the biomolecules to dictate the structural feature in the resulting nanoparticle and protein composites [4,5].
Magnetic-controlled bioelectronics is a rapidly progressing interdisciplinary research field that comprises the development of biosensors, biofuel cells, and bioelectronic devices [6]. Herein, we synthesized a magnetic-core/porous-shell CoFe2O4/Cu2O composite nanoparticles for the control of protein-nanoparticles. Cu2O, a semiconductor that displays multiple excitons in the visible spectrum, is an excellent model material for studying protein-induced nonequilibrium phases, because the thermodynamics of phase formation in the copper-water-chloride system is very well established [2,7]. The porous Cu2O matrix possesses physical rigidity, chemical inertness, high photochemical, biodegradational and thermal stability, and experiences negligible swelling in both aqueous and organic solutions, thus, and results in high protein loading and retains the bioactivity of the immobilized protein [8]. The aim of this work is to design a new immunosensor for the detection of hepatitis B surface antigen, as a model, in clinical immunoassays. This immunosensor was fabricated using chemically functionalized magnetic composite nanoparticles as immunosensing probes, and quartz crystal microbalance was employed for the measurement protocol.
2. METHODOLOGY
Initially, we synthesized the CoFe2O4 nanoparticles according to the literature [9]. Briefly, Fe(NO3)3·9H2O, Co(NO3)3·6H2O, and Glycine (Gly) were dissolved in distilled water (Fe3+/Co2+ = 2/1, Gly/nitrate = 4/1, in molar ratio). After filtration, the attained red precursor solution has been heating until combustion reaction was appeared. The black loose powders (i.e. CoFe2O4 nanoparticles) were obtained after combustion for several seconds. Following that, 0.5 g CoFe2O4 was added into a mixed solution containing 5 mL ethanol, 90 mL deionized water, 30 mg cetyltrimethylammonium bromide (CATB) and 5.0 g NaOH. After stirring for 30 min at room temperature, 2.5 g CuSO4·5H2O aqueous solution (50 wt.%) was dropped slowly, and continuously stirred for 2 h. Finally, the mixture was centrifuged, washed, and dispersed in deionized water. The mean sizes were 30 and 50 nm for CoFe2O4 and the CoFe2O4/Cu2O nanoparticles by TEM, respectively (Fig.1 ).
).
 |
Fig. (1) TEM micrographs of (a) CoFe2O4 nanoparticles and (b) magnetic-core/porous-shell CoFe2O4/Cu2O nanoparticles. |
All gravimetric measurements were performed by a quartz crystal microbalance with AT-Cut, 10 MHz quartz crystals and a gold plated electrode (diameter in 13.7 mm, 1000 Å thickness) on both sides mounted in a HC6/U holder (QCM, Pico Balance, Italy). The principle of the QCM sensors is based on changes Δfx in the fundamental oscillation frequency on the antigen-antibody interaction. To a first approximation the frequency change Δfx results from an increase in the oscillating mass Δm according to the Sauerbrey equation [9]:
where Δfx is the resonant frequency difference (Hz); f0 is the basic resonant frequency of the crystal (MHz); Δm is the mass accumulation on the crystal surface (g). A is the deposited electrode area (1.47 cm2). The frequency shift, Δfx (Hz), was defined as the absolute value of the frequency difference Δfx.
The prepared QCM probe was first mounted one side of the detection vessel containing an assay buffer solution (PBS, pH 7.4). Each of the samples to be analyzed was then introduced into the detection vessel after stabilization of resonance frequency (shift less than 1 Hz/min). To avoid the possible error resulting from different additions of samples and deduct the response induced by nonspecific adsorption, the frequency changes were recorded as the immunoreaction proceeded from 30 s (after the addition of samples) until equilibrium. The frequency changes in all experiments were referred to the average responses of immunoreaction with corresponding standard deviations (Δfx+ SD) of triplicate measurements, unless otherwise indicated. All measurements were conducted at room temperature.
3. RESULTS AND DISCUSSION
To further investigate the formation of the magnetic-core/porous-shell CoFe2O4/Cu2O nanoparticles, we used N2 adsorption-desorption isotherm to measure the specific surface area and pore size distribution of the powdered materials. The dry samples were evacuated and cooled to a temperature of 77 K, the temperature of liquid nitrogen. At this temperature inert gases such as nitrogen will physically adsorb on the surface of the samples. This adsorption process could be considered to be a reversible condensation or layering of molecules on the sample surface during which heat is evolved. In the range of 0.7–1.0 Pa, step like curves were due to capillary condensation taking place in porous material. Seen from (Fig.2 ), BET surface area and the pore size with BJH diameter of the synthesized magnetic-core/porous-shell CoFe2O4/Cu2O nanoparticles were 637.2 m2/g and 2.3 nm, respectively.
), BET surface area and the pore size with BJH diameter of the synthesized magnetic-core/porous-shell CoFe2O4/Cu2O nanoparticles were 637.2 m2/g and 2.3 nm, respectively.
 |
Fig. (2) N2 adsorption-desorption isotherms at 77 K for the magnetic-core/porous-shell CoFe2O4/Cu2O samples (Inset: Pore size distribution). |
To explore the application of the synthesized magnetic-core/porous-shell CoFe2O4/Cu2O nanoparticles in analytic biochemistry, the synthesized nanoparticles were employed as the support carrier for the immobilization of hepatitis B surface antibodies (HBsAb), as a model protein. The detection method is as follows. Initially, the synthesized nanoparticles were immobilized onto the surface of quartz probe with the aid of external magnet (saturation), and then HBsAb samples with various concentrations were injected and assayed by using quartz crystal microbalance (QCM) technique, respectively. The immobilized amount of HBsAb was evaluated by using the frequency shift before and after the adsorption according to the Sauerbrey equation [10]. Seen from (Fig.3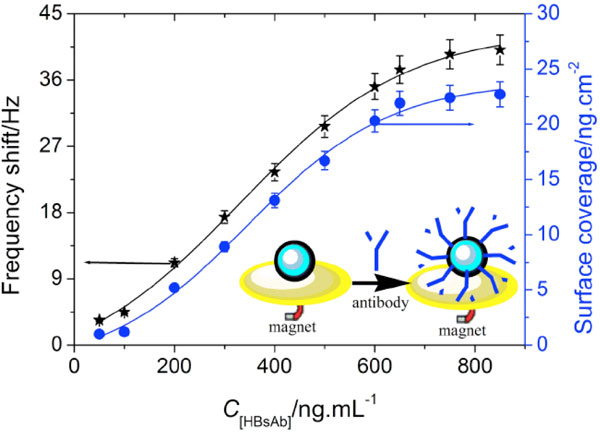 ), the frequency shifts were increased with the increment of HBsAb concentrations in the sample solution, and tended to the equilibrium at 650 ng/mL HBsAb (i.e. the maximum surface coverage was 21.9 ng/cm2).
), the frequency shifts were increased with the increment of HBsAb concentrations in the sample solution, and tended to the equilibrium at 650 ng/mL HBsAb (i.e. the maximum surface coverage was 21.9 ng/cm2).
To investigate the possibility of the immobilized HBsAb on the surface of magnetic-core/porous-shell nanoparticles in the clinical immunoassays, the HBsAb-functionalized nanoparticles with 650 ng/mL HBsAb were employed for the detection of hepatitis B surface antigen (HBsAg) by using QCM technique. Seen from (Fig.4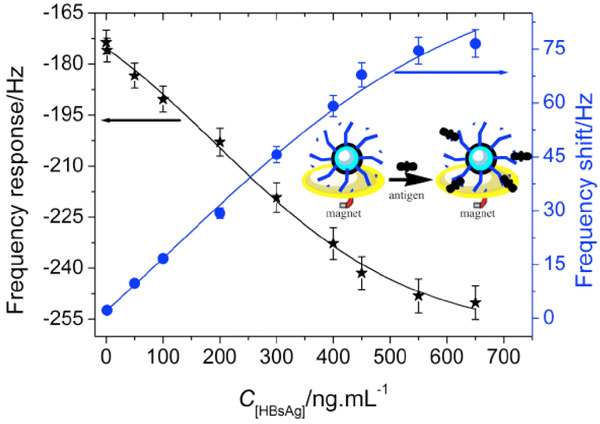 ), the frequency responses decreased with the increment of HBsAg concentration, while the frequency shift increased with an increasing HBsAg concentration. The frequency shifts were proportional to HBsAg concentration in the range of 1.5 – 450 ng/mL with a detection limit of 0.5 ng/mL (estimated to be 3 × the standard deviation of zero-dose response) (R2 = 0.991).
), the frequency responses decreased with the increment of HBsAg concentration, while the frequency shift increased with an increasing HBsAg concentration. The frequency shifts were proportional to HBsAg concentration in the range of 1.5 – 450 ng/mL with a detection limit of 0.5 ng/mL (estimated to be 3 × the standard deviation of zero-dose response) (R2 = 0.991).
 |
Fig.(4) QCM responses of the immunoprobe towards various HBsAg concentrations. |
The bionanoparticles exhibited satisfactorily stability. In fact, as much as 90.2% of the initial frequency response was preserved after storage of the nanoparticles in pH 7.4 PBS for 19 days.
4. CONCLUSION
In summary, the synthesized magnetic-core/porous-shell CoFe2O4/Cu2O nanoparticles could be employed as the immobilized matrixes for the adsorption of proteins. The potential of this method for application is a simple and efficient. Importantly, the results are preliminary and that new experiments better representing physiological conditions are planned to consolidate and validate such biosensors.

